(Funding
from DOE Grant # DE-EE0003129)
HYDROGENATION
OF AROMATICS AND HETEROAROMATICS BY SUPPORTED METAL
NANOPARTICLES
Fossil
fuels—coal, oil and natural gas—currently provide over
80% of the energy consumed in the US and will continue
to be the predominant energy source worldwide by 2035,
in spite of the aggressive development of alternative
renewable energy sources. (U.S. Energy Information
Administration, Washington, DC, 2010, 2011.
International Energy Agency, Paris, France, 2010).
Environmental regulations impose increasingly severe
restrictions on the aromatics, sulfur, and nitrogen
content allowed in fuels and the low levels required are
difficult to achieve with current refining technologies.
Catalytic hydrogenation plays a pivotal role in lowering
the proportion of aromatic hydrocarbons (HYD), and in
removal of nitrogen and sulfur through
hydrodenitrogenation (HDN) and hydrodesulfurization
(HDS). Conventional metal sulfide hydrotreating
catalysts (Co-Mo, Ni-Mo and Ni-W) saturate only a
moderate proportion of aromatics due to thermodynamic
equilibrium limitations under operating conditions.
Supported noble metal catalysts can function at lower
temperatures far from equilibrium conditions, but they
are easily poisoned by N- and S-containing compounds
present in refinery feeds. Therefore, there is a
continuing need for catalysts capable of promoting
hydrogenation of aromatics, while being resistant to
poisoning by N- and S-species.
Our working hypothesis is that a surface nanostructure
composed of small metallic particles immobilized on a
support that is rich on basic sites can promote rare
heterolytic hydrogen splitting and ionic hydrogenation
mechanisms through a metal-support bifunctional effect,
thereby avoiding strong substrate adsorption to active
sites that leads to catalyst poisoning:
-
To develop novel nanostructured materials specifically designed to promote the heterolytic splitting of molecular hydrogen and ionic hydrogenation mechanisms, by use of metallic nanoparticles immobilized on solid supports containing strongly basic functionalities
-
To optimize their catalytic properties in the hydrogenation of aromatics and heteroaromatics representative of components of petroleum-derived fuels, and the dehydrogenation of N-heterocycles, of possible utility as liquid hydrogen storage materials, and to understand the principal reaction mechanisms
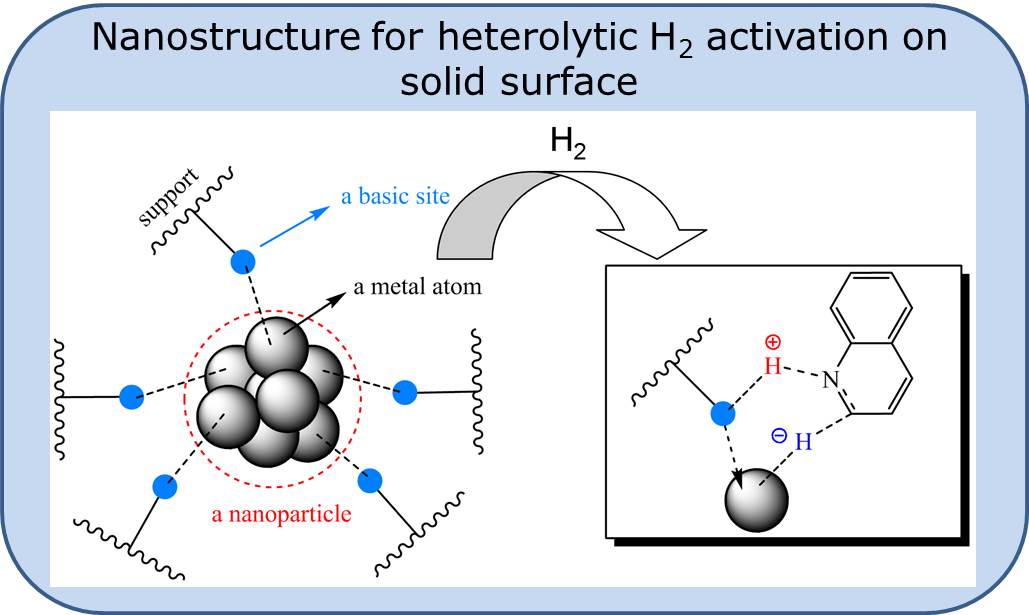
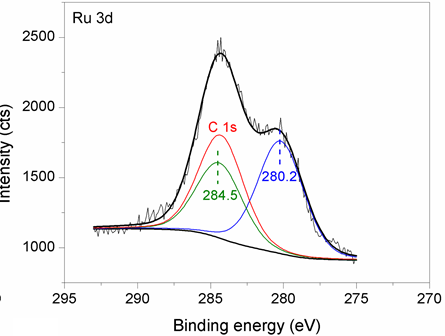
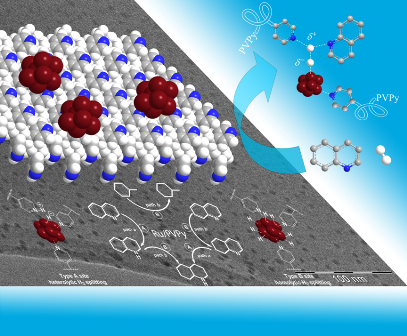
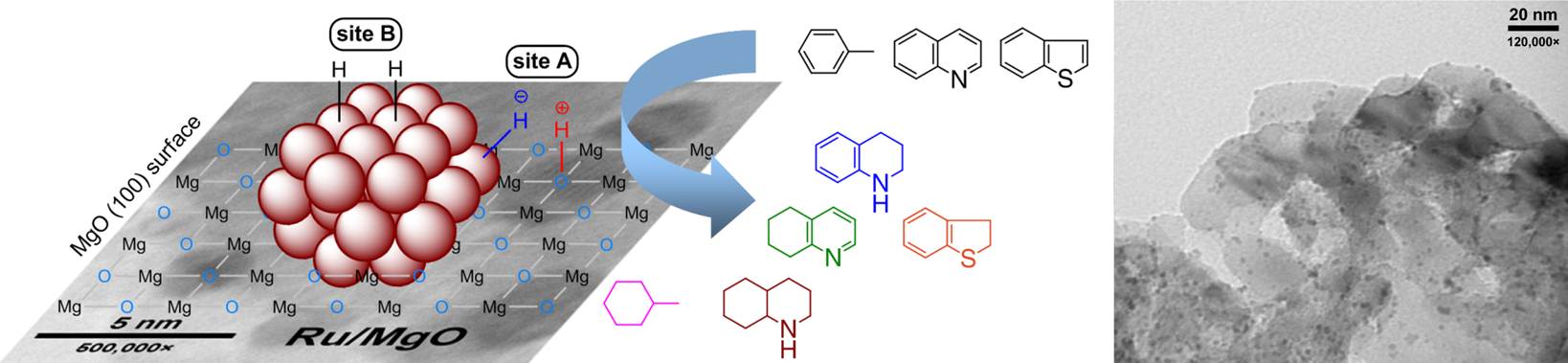
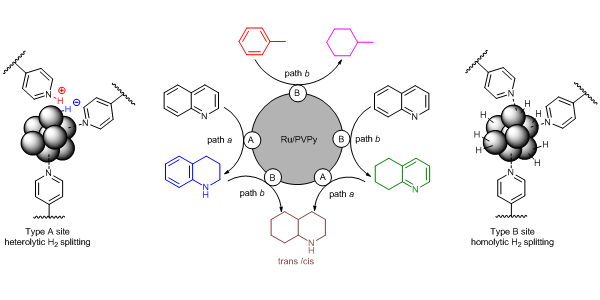
The catalyst operates through a novel dual-site substrate-dependent mechanism.
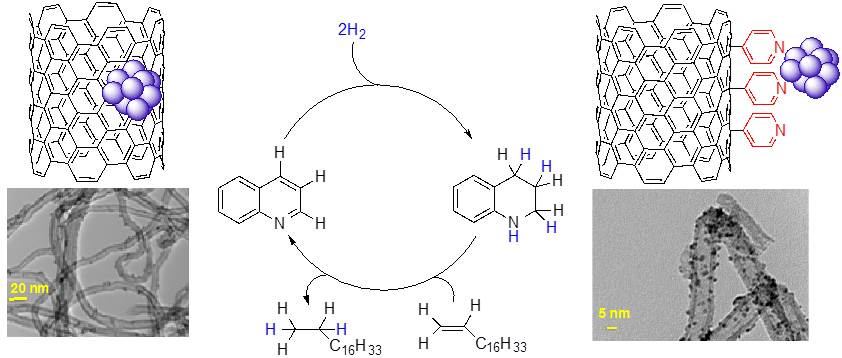
Hydrogen storage in organic liquids: Hydrogenation-dehydrogenation of N-heterocycles by Ru nanoparticles supported on functionalized carbon nanomaterials.
Functionalized multiwalled carbon nanotubes, and graphene oxide are being employed as catalyst supports for Ru nanoparticles.
The new materials hydrogenate a variety of arenes and N-heterocycles under moderate reaction conditions and also promote the mild dehydrogenation of saturated N-heterocycles. The hydrogenation-dehydrogenation couple represents an interesting alternative for hydrogen storage in organic liquids.
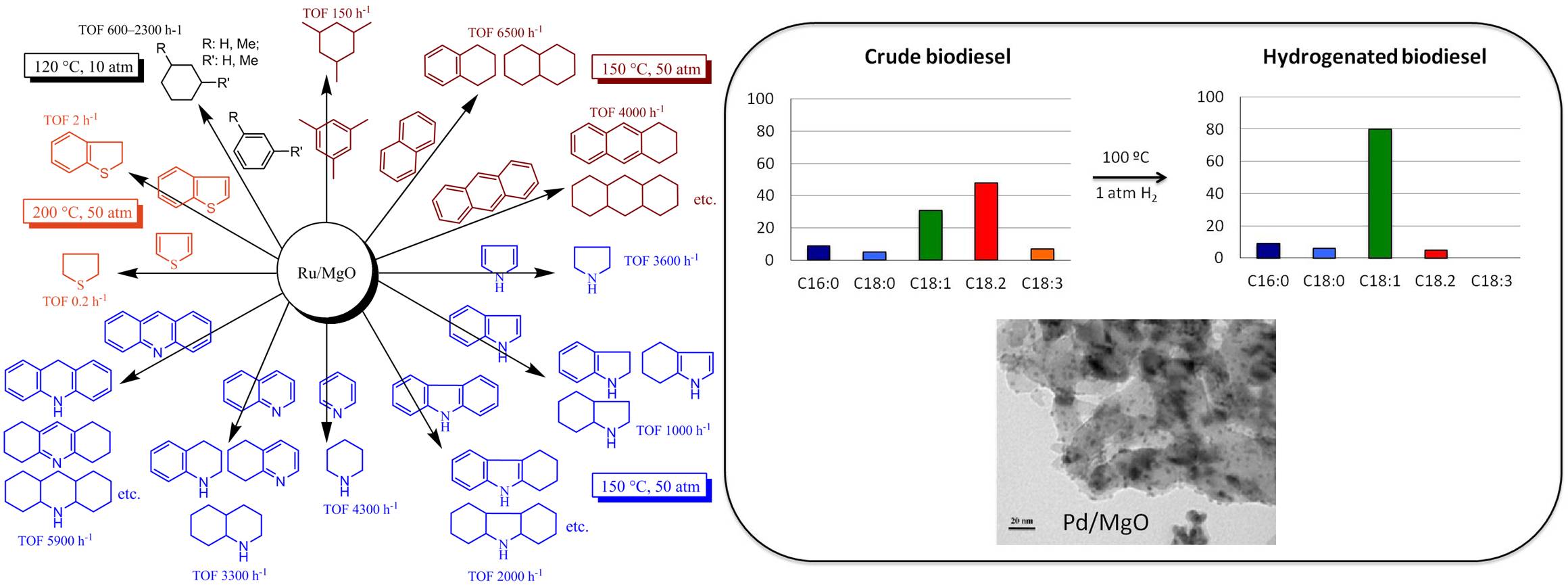
Pyrolysis of lignocellulosic biomass (wood, agricultural waste, grass) produces bio-oil, which displays poor combustion properties mainly due to its high oxygen content. Catalytic hydrotreating is needed to remove oxygen and produce liquid hydrocarbons. Supported metal sulfide catalysts used in petroleum hydrotreating or supported noble metals can be applied to bio-oil but they are not efficient enough for industrial applications. Therefore, new effective HDO catalysts are needed.
A key step in HDO is the cleavage of C-O bonds but due to the intrinsic complexity of the solid catalysts and surface reaction networks involved, crucial mechanistic issues remain unsolved. Our aim is to understand the factors that influence organometallic reactions leading to selective C-O bond scission of bio-fuel components, and the mechanistic pathways by which such processes take place. We study the interactions of transition-metal complexes of tripodal P- and mixed P,N-donor ligands with model substrates by experimental and theoretical methods.
-
 M. Fang and R. A.
Sánchez-Delgado, “Ruthenium nanoparticles supported
on magnesium oxide: a versatile and recyclable
dual-site catalyst for hydrogenation of mono- and
poly-cyclic arenes, N-heteroaromatics, and
S-heteroaromatics” 2013 (submitted)
M. Fang and R. A.
Sánchez-Delgado, “Ruthenium nanoparticles supported
on magnesium oxide: a versatile and recyclable
dual-site catalyst for hydrogenation of mono- and
poly-cyclic arenes, N-heteroaromatics, and
S-heteroaromatics” 2013 (submitted) -
A. Sánchez, M. Fang, A. Ahmed, and R. A. Sánchez-Delgado, “Hydrogenation of arenes, N-heteroaromatic compounds, and alkenes catalyzed by rhodium nanoparticles supported on magnesium oxide” 2013 (submitted)
-
R. Rahi, M. Fang, A. Ahmed and R. A. Sánchez-Delgado, “Hydrogenation of quinolines, alkenes, and biodiesel by palladium nanoparticle supported on magnesium oxide.” Dalton Trans. 2012, 41, 14490-14497.
-
M. Fang, N. Machalaba and R A. Sánchez-Delgado, “Hydrogenation of arenes and N-heteroaromatic compounds over ruthenium nanoparticles on poly(4-vinylpyridine): a versatile catalyst operating by a substrate-dependent dual site mechanism” Dalton Trans. 2011, 40, 10621–10632.
-
S. Jiménez, J. A. López, M. A. Ciriano, C. Tejel, A. Martínez, and R. A. Sánchez-Delgado, “Selective Hydrogenation of Cinnamaldehyde and Other α,β-Unsaturated Substrates Catalyzed by Rhodium and Ruthenium Complexes” Organometallics, 2009, 28, 3193-3202.
-
R. A. Sánchez-Delgado, “Organometallic Models of the Hydrodesulfurization and Hydrodenitrogenation Reactions”, Catalysis by Metal complexes Series, Nº 24, Kluwer Academic Publishers, Dordrecht, The Netherlands (2002)