MINERALS
Authors: David Leveson and David Seidemann
Edited for the web: Guillermo Rocha
Rocks and minerals are part of our daily experiences. If we look around us we are surrounded by objects that are made of rocks and minerals. For example, metal cabinets are made of alloys of different types; the circuit boards in computers, television sets, radios, dvd players, etc. are made of minerals. Homes are made of bricks, sheet rocks, stone and cement all these components made of rocks. Piping, electrical wiring, porcelain bath tubs and toilets all made minerals. Cars, planes, boats, bicycles, motorcycles, trains, etc. all made from minerals extracted from rocks. Highways, bridges, tunnels are made on the foundation of different types of rocks. The examples of objects that are made of minerals and rocks are endless.
Earth is composed of the approximately 100 naturally occurring chemical elements in a vast number of combinations. However, 98.5% of Earths crust is composed of only 8 elements: oxygen (O), silicon (Si), aluminum (Al), iron (Fe), calcium (Ca), sodium (Na), potassium (K), and magnesium (Mg). In materials such as gases, liquids, and glass atoms may occur in random proportions, with the atoms having no fixed relationship in space to each other (Fig. 1A). However, in most solids the elements combine in fixed proportions, with the atoms occupying definite positions in space to form crystal lattices.
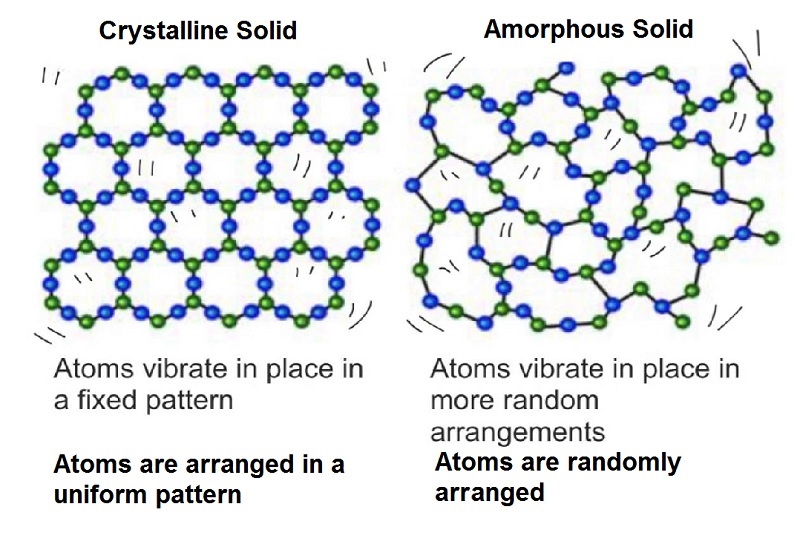
By definition a mineral is a naturally occurring homogeneous inorganic crystal having a definite chemical composition and characteristic crystalline structure, color, and hardness.
For substance to be called a mineral must have these 4 requisites:
1) must be solid
2) must be inorganic
3) must be naturally occurring
4) must be crystalline
Properties of minerals - Each mineral is unique and each has a distinct chemical and physical properties. These physical and chemical properties depend upon a) which elements they contain, and b) the arrangement of these elements in space.
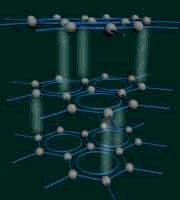
For example graphite and diamond have the same exact chemical composition (pure carbon), but because of their atomic arrangement:
 |
 |
|
|
Diamond: extremely hard, brilliant and transparent. |
||
 |
 |
|
| Graphite: extremely soft, dull and metallic. | ||
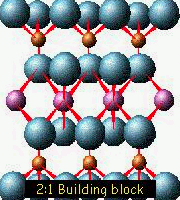
There are some minerals that have a almost similar atomic structure but a different chemical composition, then their properties also change:
 |
 |
||
|
Muscovite: No iron in its structure, colorless, transparent and an excellent insulator. |
|||
 |
 |
||
|
Biotite: Iron on its structure, black and a conductor of electricity. |
|||
Difference between a rock and a mineral

In order to study minerals without sophisticated equipment, we look at the following characteristics:
5. Reaction to Hydrochloric Acid
©David Leveson and David Seidemann - Brooklyn College - Earth and Environmental Sciences