|
1. You have been given an unknown mineral called 'C'.
- Determine its weight to the nearest tenth of a gram. To do this, examine Fig. 1 shown below. It shows the scales of a triple beam balance, calibrated in grams.
The weight is: 538.7 g
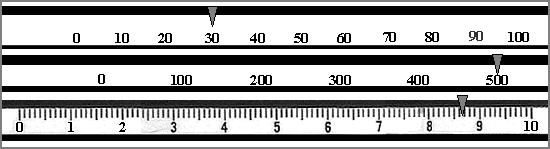
FIGURE 1
- Next, determine the volume of mineral C to the nearest 10 cubic centimeters. To do this, examine Fig 2 shown below. Fig 2A shows the level of water in a graduated cylinder (calibrated in cubic centimeters) before the mineral is immersed. Fig. 2B shows the water level after the mineral is immersed.
The volume is 830-690 = 140 cc

FIGURE 2A | | 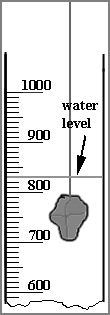
FIGURE 2B |
- Now, calculate the density of mineral C.
The density is 539/140 = 3.85 g/cc
- Last, identify the mineral, using Table 1.
The mineral is siderite.
2. Both mineral X and mineral Y have the following properties: -
color = blue;
- density = 2.65 g/cc.
Consult Table 1 to answer these questions:
What two minerals might they be? cordierite or quartz.
What test might distinguish them? A test for hardness.
3. Mineral Q has the following properties: - It scratches calcite and is scratched by orthoclase.
- It has no streak.
- Its density is less than 3.
- Its color is white.
Consult Table 1 to answer this question: What three minerals might it be?
heulandite
aragonite
apophyllite
Describe a specific test that could be applied to Q that might tell you (a) which of the three minerals it is, or (b) might tell you which of the three minerals it is not.
The test would be to: see if the mineral scratches fluorite (H=4).
Questions 4 to 10. Review the "What is..? and "What are..?" Lou-Lulu dialogs.
| 
