| Crystalline substances can be formed artificially in controlled laboratory experiments. Ways in which they can be formed include from cooling solutions, evaporating solutions, cooling melts, and substances that are annealed (heated - but not melted - to alter physical properties such as strength and hardness). Information gained from such experiments may be relevant to the formation and grain size of crystalline substances in nature. |
In this experiment, a solution is shown cooling over a period of three hours. Initially, there are no crystals. After one hour, a crystal has formed and has grown to the size shown. The crystal continues to grow during the remaining two hours. To review what is happening at an atomic level, visit 'Attachment By Intergrowth'.  | 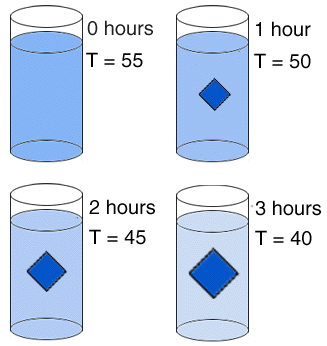 |
| In this experiment, the solution is cooled more rapidly over the same length of time. At the end of one hour, three crystals have formed, but each is smaller than the single crystal that formed in the first experiment. Over the remaining two hours, the three crystals continue to grow, but never attain the size of the single crystal. | 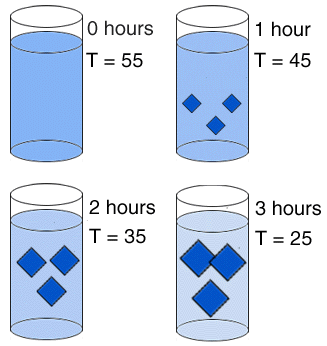 |
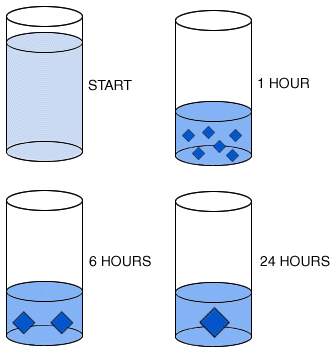
| In this experiment, a solution in a cylinder is evaporated down to about 40% of its starting volume over a period of one hour. As the volume of the solution diminishes, crystallization occurs. In a second run of the experiment, evaporation of the same volume of liquid occurs over six hours. In a third run of the experiment, evaporation of the same volume of liquid occurs over 24 hours.
 Question 1: Describe the relationship between the rate of evaporation, crystal size, and number of crystals. Question 1: Describe the relationship between the rate of evaporation, crystal size, and number of crystals.
|
|  |
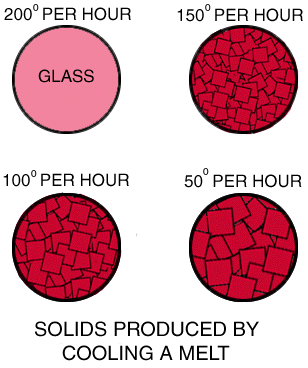
In this experiment, four temperature controlled containers of the same size are filled with identical melts. All the melts are allowed to cool until they solidify completely. However, the rate at which each melt is allowed to cool differs. One is allowed to cool 200 degrees per hour; the others are allowed to cool 150, 100 and 50 degrees per hour. At the fastest rate of cooling (200 degrees per hour), no crystals formed. The melt was frozen into a glass. At slower rates of cooling, crystals formed.
 Question 1: As the cooling rate is decreased, what happens to the size of the crystals?: Question 1: As the cooling rate is decreased, what happens to the size of the crystals?:
 Question 2: As the cooling rate is decreased, what happens to the number of crystals? Question 2: As the cooling rate is decreased, what happens to the number of crystals?
|
|  |
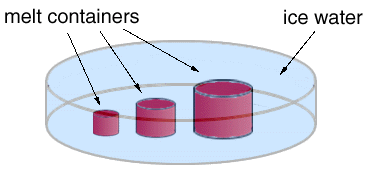
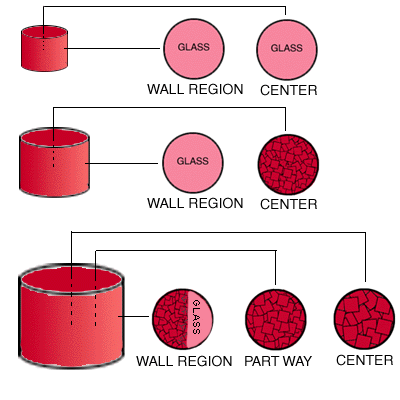
In this experiment, three insulated, cylindrical containers of different size are filled with identical melts at the same initial temperature. All the containers are then immersed in a large vat of ice water whose temperature is maintained at zero degrees Centigrade (32 degrees Fahrenheit). The containers are kept in the ice water until all the melts have cooled and solidified completely. Samples of the solidified melts are then taken from the centers and and wall regions of the two smaller containers, and from the center, wall region, and a point halfway between the center and the wall region of the largest container. The samples are shown in the circles to the right of the cylinders.
 Question 1: Why did the melt in the smallest container turn completely into glass?: Question 1: Why did the melt in the smallest container turn completely into glass?:
 Question 2:Why did glass form only near the wall region of the intermediate size container but not at its center?: Question 2:Why did glass form only near the wall region of the intermediate size container but not at its center?:
 Question 3: At the wall region in the largest container, why did only a very thin layer of glass form?: Question 3: At the wall region in the largest container, why did only a very thin layer of glass form?:
 Question 4: Describe and explain the variation in grain size from the center out to the wall region of the largest container.: Question 4: Describe and explain the variation in grain size from the center out to the wall region of the largest container.:
|
|  
|
| In this experiment, a solid metal cylinder is heated until it is red hot and then allowed to cool. A comparison of the texture before and after heating shows that the grains have changed shape and size. | 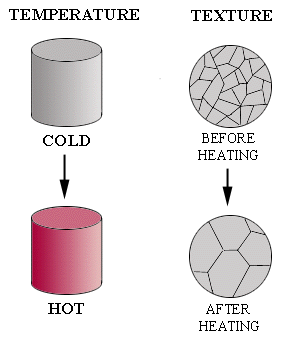 |
To check your answers and get additional hints and information, click on the link. |