| |||
| In this experiment, a drop of concentrated salt water is placed on a microscope slide and is allowed to evaporate. The sequence of crystallization events that accompany evaporation may be viewed through a microscope. Salt forms cubic crystals which here are represented as squares or rectangles. The area putlined in red will be examined.
Stage 1: A row of small square crystals forms along the edge of the drop. Stage 2: Rows of progressively larger square crystals form along the margins of the drop. In the interior of the drop, isolated square crystals begin to form. Stage 3: Rows of progressively larger square crystals continue to form along the margins of the drop. The isolated square crystals in the interior increase in size. Stage 4: The square crystals in the interior grow so large that they begin to impinge on one another.
Stage 5: Myriads of small crystals form in the spaces between the large interior crystals. Few of the small crystals are perfect squares. Most are parts of squares; some have no elements of 'squareness'. Stage 6: All the grains develop a corroded, worm-eaten appearance. | 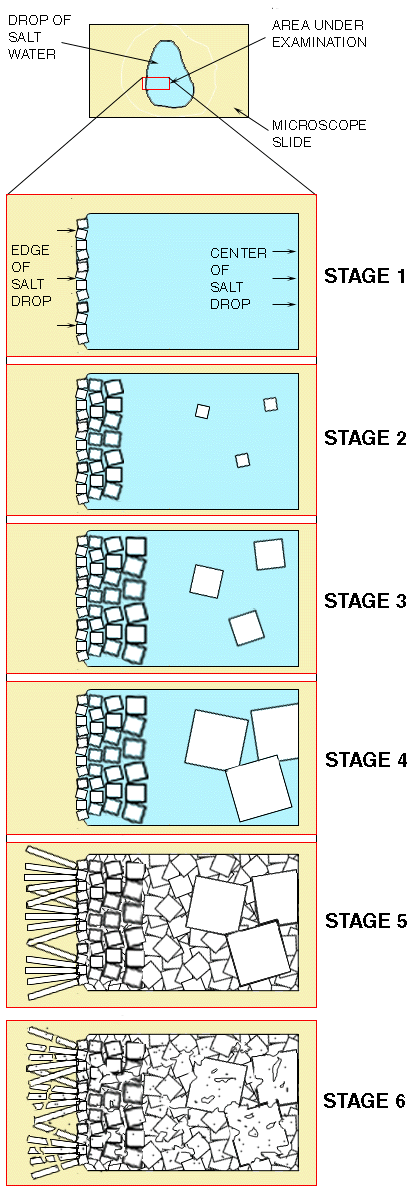 |
||
The shapes of the grains of salt that crystallized from the salt solution vary in several ways:
| |||