

|
| In radioactivity a single nucleus breaks into two parts. In other nuclear processes, two nuclei come close together, exert forces on one another, and one or both undergo a change. This is called a nuclear reaction. | |
| Conditions for Nuclear Reactions | If two nuclei are surrounded by electrons, as they are under ordinary conditions on earth, it is not possible for a nuclear reaction to occur. This is because the electron clouds of the two nuclei repel each other since the electrons all have negative electric charge. Two atoms might "bounce off" each other or might hold together to form a molecule, but in either case the nuclei remain separated by a distance of about 10-10 m. Since the nuclear force only reaches a distance of about 10-15 m, neither nucleus can be influenced by the other. So to have a nuclear reaction at least one of the nuclei must be "naked", have all its electrons removed. This can be accomplished by bringing matter to very high temperatures (at least 100,000 degrees). |
| High energy needed | Now the naked nucleus A can plow through the electron cloud of atom B and approach nucleus B. But there is another problem. The two nuclei are both positively charged and repel each other. This electrical repulsion acts over long distances, and may prevent A and B from getting close enough together to interact strongly. The electrical repulsion can be overcome if the nuclei approach each other at high speed. There are three ways that can happen:
A beam of fast particles, say, oxygen nuclei, approaches a target, perhaps a solid piece of carbon. Many of the beam particles go right through the target (through the electron clouds between the carbon nuclei) and come out the other side without having come close to any carbon nuclei. Some come close to a carbon nucleus but just bounce off in some other direction. Others undergo nuclear reactions.
|
| Here are some examples of nuclear reactions: | |
|
Transfer Reactions |
Suppose the beam consists of the rare oxygen isotope, 178O. The following is called a transfer reaction: |
 | |
| One of the 9 neutrons on the oxygen has transferred to the carbon. Another example: suppose you had a beam of heavy hydrogen, 21H, and a target of neon, the common isotope, 20Ne. The transfer reaction, | |
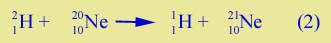 | |
|
might occur. Again a neutron is transferred.
| |
| Transmutation | It's also possible for a proton to be transferred from one nucleus to another:
|
 | |
|
Note here that when the nitrogen nucleus loses a proton, it is left with 6 protons and 7 neutrons. Therefore it can no longer be called "nitrogen" because its atomic number is now 6. The atomic number defines the chemical element, and it now must be called "carbon". Similarly, when 21H receives a proton it becomes a nucleus with atomic number 2; it is helium.
| |
| Constant proton and neutron numbers |
You can check the correctness of any of these nuclear reactions by noting that the total mass number is the same before and after the reaction; also the total atomic number is the same. Protons and neutrons are not created or destroyed; they are just shifted around. Reaction (3) represents a change of one element into another, what used to be called "transmutation of the elements". The alchemists attempted to change ordinary metals in gold but never succeeded. Later the principle that elements were immutable in chemical reactions was recognized. In nuclear reactions elements do change. Lead has been changed into gold. (Reaction (3) shows the production of a carbon-13 nucleus. Later that nucleus will gather to itself 6 electrons and become a carbon atom. A large collection of these atoms will be a piece of carbon.) Here is a more complicated transfer reaction leading to a new element:
|
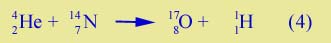 | |
| Three nucleons from the helium nucleus (one proton and two neutrons) get transferred to make the oxygen. This process occurred in the first transmutation experiment, done by Rutherford in 1919, although it was not fully understood until a few years later. | |
| Discovery of the neutron |
Another example:
|
 | |
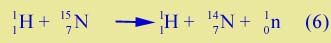
| Three particles are transferred from the helium to the beryllium (what are they?), leaving a free neutron and carbon-12. The neutron might be represented by 10n, since it has zero protons and mass number 1. Reaction (5) is the one in which the neutron was first discovered by Chadwick in 1932. It is also the basis of the neutron generator which we use in the lab on radioactivity. The helium-4 particles come from radioactive americium, and the beryllium is just a piece of metal. Inside the large barrel of water free neutrons are produced. | |
|
Knock-out Reactions |
A nuclear reaction may result in more than two products, the following is called a knock-out reaction: |
 | |
|
Neutron Absorption | Sometimes two nuclear particles stick together. The following reactions are called neutron absorption:
|
 | |
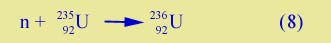 | |
|
27Al is the common isotope of aluminum; 235U is a rare isotope of uranium. The products, aluminum-28 and uranium-236, are both very unstable and not found in nature . (For that reason 236U is not listed in your nuclear table.) Neutron absorption is important in a nuclear reactor, where control rods, made of materials that readily absorb neutrons, keep fission going at a steady rate.
| |
| The neutron generator | Aluminum-28 is the nucleus you will study in the lab on radioactivity. The experiment begins when the instructor places a disk of ordinary aluminum (27Al) inside the neutron generator. Neutrons produced via reaction (5) hit the disk, and some of them get absorbed by aluminum nuclei to produce the artificial nucleus, 28Al. There is a reason why absorption processes like (7) and (8), are done with neutrons. In the discussion above we said that if two nuclei are to come together and undergo a reaction, they have to be moving fast in order to overcome the electric repulsion. If we start with a neutron there is no electric repulsion between it and any nucleus. Therefore we can initiate a reaction with neutrons even if they are moving slowly. This turns out to be important in nuclear reactors. |
| Slow Neutrons |
Another important point about neutron absorption is that the process depends strongly on the speed of the neutrons. Neutron absorption is much more likely to occur if the speed of the neutrons is low. At higher speeds (like the speeds of radioactive particles) a neutron hitting a nucleus is more likely to just bounce off, causing the nucleus to recoil. It is a little like trying to catch a rubber ball with one hand. At high speeds the ball might hit your palm and bounce off, but a slower ball may just sink into your hand. |
|
Trans-uranic Elements | During the 1930's a great deal of experimental work was done with neutrons, because nuclear reactions can be induced even with slow neutrons. One of the objects was to produce new (artificial) elements with atomic number higher than Z = 92, uranium. If 238U absorbs a neutron,
|
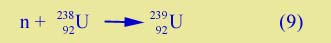 | |
|
the new isotope, 239U, is more unstable than 238U (remember 238U is very slightly unstable; it undergoes radioactive decay, but very slowly). 239U can beta decay,
| |
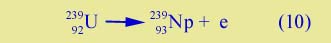 | |
|
The element with Z = 93 is not found naturally (on earth, or elsewhere in the universe). It is created for the first time in this experiment. It is called neptunium. Neptunium itself beta decays,
| |
 | |
|
producing another artificial element, plutonium.
| |
| | |
| At this point you should begin to work on Problem Set 5. | |
| |
|
| In a similar way, the other elements beyond uranium are created by neutron absorption, followed by one or more beta decays, leading to nuclei with more protons - higher atomic number. These are all radioactive. | |
|
Scattering | Another important nuclear process, which strictly speaking doesn't fall under the category of nuclear reactions, is called "scattering". In this process, two nuclei come together and exert strong forces on each other, but there is no rearrangement of neutrons and protons. The two nuclei are unchanged, but their velocities after the interaction are different than what they were before. Typically it might look like a collision on a pool table: One nucleus is stationary, the other comes in and "hits" it, and then the two move off at different angles. In this process, the incoming nucleus loses some of its energy. This is important in the action of the "moderator" in the design of nuclear reactor. |
KEY CONCEPTS
|