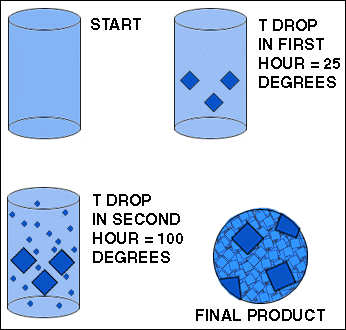
| In this experiment, a melt is shown cooling over a period of several hours. Initially, there are no crystals. During the first hour, the temperature drops 25 degrees. Several crystals form. During the second hour, the temperature drops 100 degrees. The crystals that formed during the first hour are joined by a host of smaller crystals. Cooling then continues at the same rate (100 degrees per hour) until the entire melt is crystallized. The resulting mixture of large and small crystals is shown in the circle on the lower right.
Similar results are obtained for cooling or evaporating solutions in which the rate of cooling (or evaporation) is increased partway through the crystallization process.
Experiments to determined what controls grain size uniformity when crystalline grains form within a solid are difficult to perform and to interpret. They will not be considered.
|  |