

|
| Discovery
|
In 1939 Otto Hahn and Fritz Strassman, experimenting with neutrons and a pure sample of uranium, found small traces of the element barium in the sample. Hahn and Strassman were chemists, and in the tradition of Marie Curie would not have been surprised to discover new elements produced by radioactivity (like thorium). But the elements usually found in this way have high atomic numbers, close to that of uranium. Barium is Z = 56. This suggested that a uranium nucleus has split into two parts; but unlike normal radioactivity, where one part is very much smaller than the other (e.g. an alpha particle has Z = 2, thorium has Z = 90), here the uranium is split into two approximately equal parts: the second piece must have Z = 92 - 56 = 36. Element number 36 is krypton, a rare gas. |
 | |
|
The mass numbers at first were unknown. Barium was identified via its chemical properties. And krypton was not there: since it's a gas, it would have just drifted up into the air - not stayed in the uranium sample. This process was called fission, in analogy with the method of reproduction of one-celled animals.
| |
| Isotopes of U
|
Natural uranium contains the two isotopes, 235U and 238U. For reasons to be discussed below, we will consider the example of fission of 235U. Since fission only occurs when neutrons are present, what really happens is that 235U first absorbs a neutron. The resulting 236U is very unstable, and instead of decaying via some already known form of radioactivity, it decays via fission. |
| Free Neutrons |
Suppose we hypothesize that the Ba and Kr isotopes in process (1) are the most common naturally found isotopes. Then (1) becomes,
|
 | |
|
There is something wrong with this. If you add up the number of neutrons you find there are 14 missing on the right side. Where are they? It may be that we really have different Ba and Kr isotopes than the ones given above. Or it may be that in addition to the two medium-sized nuclei produced, there are also some free neutrons released in the fission process. Around 1940 an important experiment was done (at Columbia University) to determine if free neutrons were emitted in fission, and how many. The result was that on the average about 3 neutrons were released per fission. (The reason we say "on the average" is that fission is occurring in a large number of uranium nuclei, and we have different numbers of neutrons released in different cases, but the average number is 3.) Thus a typical fission process might be the following sequence:
| |
 | |
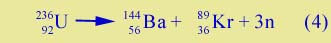 | |
| Why are extra neutrons produced? |
The appearance of extra neutrons is crucial to the matter of nuclear energy, as we will see. Why does this happen? Recall that as you go to heavier and heavier nuclei in the nuclear table the number of neutrons relative to protons gets larger and larger. Therefore if a heavy nucleus like uranium splits into two medium-sized nuclei, there are too many neutrons around to fit comfortably on those two nuclei. Hence there are always a few leftover neutrons, as in Equ. (4). For a simple numerical illustration of how this works, click here. Process (4) also represents the release of a large quantity of energy. The two nuclei fly apart very rapidly. Energy is available because the total mass on the right side is less than that on the left. Although the number of protons and neutrons are the same, the krypton and barium nuclei are more tightly bound than the uranium; their binding energy is greater. |
| Fission Fragments |
Bear in mind that fission of 236U can actually occur in many different ways, and (4) is only an example. Not only can you get different barium and krypton isotopes with different numbers of free neutrons, but you can also get different elements. An example:
|
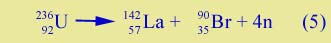 | |
|
(La is lanthanum; Br is bromine.) The medium-sized nuclei, like Ba, Kr, La, and Br, are called fission fragments. They are usually not the common isotopes of these elements. And they are usually highly radioactive.
| |
| | |
| At this point you should begin to work on Problem Set 6. | |
| |
|
| |
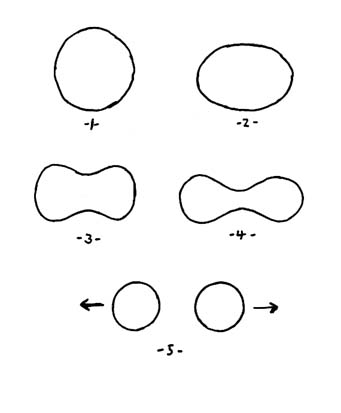
Theoretical studies during the early 1940s showed that fission involves a gradual changing of shape of the original nucleus, something like this: |
 | |
| The Chain Reaction |
Suppose you have a piece of uranium that is mostly 235U, and you introduce one neutron into it. The neutron gets absorbed by one nucleus to produce 236U, which then fissions. Following this fission there are three free neutrons. Each of these neutrons may hit another uranium nucleus and so after a time there are three more fissions, and nine free neutrons. Each of these may cause another fission , and then 27 free neutrons. There is thus a very rapid multiplying effect, so that even though we start with just one neutron, we end up with very many of the uranium nuclei undergoing fission. In a fraction of a second the entire sample, with around 1027 nuclei, can all fission. This multiplying process is called a chain reaction. |
| Why is 235U needed? |
The chain reaction exists because the average number of free neutrons in a fission is greater than 1. But there is a difference here between 235U and 238U. The number for 238U is actually slightly less than 1, while the number for 235U is greater than 3. So, while both isotopes of uranium can fission, only 235U can lead to a chain reaction. Much of the work toward building the first atomic bomb had to do with separating 235U from the natural ore (which has a mixture of both isotopes). And in the continuing concern today with proliferation of nuclear weapons, one of the chief focusses is on whether nations can produce separated 235U.
|
| Critical Mass |
When a neutron is produced in fission it does not instantaneously hit another uranium nucleus. Remember the uranium sample consists of atoms, and there are large spaces among the nuclei (occupied by electrons) through which neutrons can move without causing fission. Typically a neutron will travel 2 or 3 centimeters before hitting a nucleus. Therefore if the sample is very small (say, 1 cm in diameter), most of the neutrons will move through it and pass out the sides before causing another fission. In order for enough fissions to occur to maintain the multiplying effect of the chain reaction, the uranium sample must be a certain minimum size. If it's a sphere the minimum size turns out to be a diameter of about 20 cm. This quantity of uranium is called the critical mass. |
KEY CONCEPTS
|