

|
|
Energy and mass |
There are a number of ways of looking at Einstein's famous equation, E = mc2. E stands for energy, and the equation says that when an object has mass (m), this is a form of energy. Other forms of energy include kinetic energy (the energy of motion), heat, light and other waves. The quantity, mc2, where c stands for the speed of light, is a quantitative measure of the amount of energy in a mass, m. |
| The maximum speed |
[Although c is the speed of light, this equation doesn't really have anything to do specifically with light. The principle of relativity says that there is a maximum speed, such that nothing can travel faster than that speed. That's what "c" is - the maximum speed. Certain things travel just at c; light is one of them. Everything else is slower.] |
|
Conservation of energy |
There is a fundamental law that applies in all the sciences, which says that energy can be transferred from one form to another, but the total quantity of energy remains the same. This applies in a closed system, i.e. a system not subject to external forces. Einstein's discovery is that the energy inherent in mass can be transformed to other forms, subject to the same law, that total energy is constant. |
|
Nuclear binding energy |
There are several important ways in which Einstein's discovery comes in to nuclear physics. One is the "binding energy" of a nucleus. Take a simple example, the nucleus 21H, also called the "deuteron"; it consists of one proton and one neutron. If one measures the mass of the deuteron, it turns out to be not exactly equal to the mass of a proton plus the mass of a neutron. It is slightly less, by about 0.1%. It follows that a deuteron cannot just break apart spontaneously: |
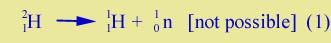 | |
| There is less energy on the left side (mass of 21H times c2) than on the right side. The deuteron can only break up if some energy is supplied to it. | |
 | |
| This energy could be supplied by another particle, say a fast-moving proton that hits the deuteron and breaks it apart. | |
 | |
| The energy that makes this possible is the kinetic energy of the incoming proton. The minimum energy needed to break apart a deuteron is called the binding energy of the deuteron. | |
|
Disintegrating a nucleus |
Similarly, for any nucleus, the binding energy is the minimum energy necessary to break it apart into completely separated neutrons and protons. Qualitatively we could say that binding energy measures how much less the mass of the nucleus is than the sum of its parts. A large binding energy means it's hard to break up the nucleus; the nucleus is tightly bound. The deuteron is an example of a weakly bound nucleus. The alpha particle (42He), and the carbon nucleus (126C) are examples of tightly bound nuclei. |
|
Attractive forces |
The underlying cause of binding energy is not mysterious: It is the existence of the nuclear force, the attractive force that holds the protons and neutrons together. If you had a dish that had been broken into three pieces which were now glued together, the dish wouldn't spontaneously break apart. It would if you dropped it on the floor (which supplies energy). |
|
Binding energy in chemistry |
There is also binding energy in chemistry. Attractive electric forces hold together two hydrogen atoms and an oxygen atom to make a water molecule, H2O. To break the molecule apart you have to supply energy - usually in the form of heat. So there is a binding energy of the water molecule. The difference between the chemistry case and the nuclear case is that the force holding nuclei together is much stronger (by a factor of about 106) than the force holding molecules together. Therefore nuclear binding energies are typically about a million times stronger than chemical binding energies. |
|
Energy release in nuclear reactions |
In a nuclear reaction in which you have several nuclei on each side of the process, each nucleus has a certain binding energy, and so there may be more total mass on the left side or more on the right side. If there is more mass on the right side (as in Equ. (1) above) the process cannot occur, unless some form of energy comes in from outside. But if there's less mass on the right, than the reaction goes, and energy is produced. The amount of energy produced is equal to the mass difference times c2. An example would be |
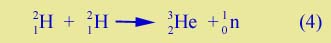 | |
| which is one of the reactions being investigated for possible use in a fusion reactor. Here the helium nucleus on the right side is more tightly bound than the deuterons on the left. In total there is more mass energy (mc2) on the left, and so energy is produced when the process goes. That energy will be in the form of rapid motion of the product particles. | |
|
Source of the sun's energy |
In the interior of the sun (and stars), nuclear reactions burn hydrogen to produce helium (the tightly bound alpha particle). Thus energy is released to heat the interior of the sun to millions of degrees, and power the emission of light and heat out into space. |
|
Fission |
In nuclear fission a large nucleus like 23692U decays into two medium-sized nuclei, e.g. |
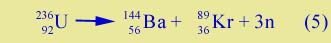 | |
| Here the binding energy of the uranium nucleus, while large, is not as large as the combined binding energies of the krypton and barium nuclei. Hence energy is released, which powers fission reactors and the atomic bomb. | |
|
Gamma rays | The energy released in nuclear reactions sometimes appears in the form of kinetic energy - fast motion of the product particles, but it also can appear in the form of gamma rays. The gamma can be thought of as a particle or as a wave, just as light can. Both are examples of the electromagnetic wave. As a particle, the gamma has a much higher energy than a particle of light (the photon). Gammas also appear as products in radioactivity. |
KEY CONCEPTS
|