

|
An atomic bomb must contain a critical mass of fissile material, that is, material that can undergo fission. Two types of fissile material have been used in atomic bombs up to the present type:
| |
|
Gun-type assembly |
The simplest method of assembling a critical mass is to begin with two hemispheres, each containing half of a critical mass, and drive them together rapidly. This is called a "gun-type" device. Chemical explosions at two ends of the bomb drive the two hemispheres together. When the critical mass (the full sphere) is formed, and a small influx of neutrons is introduced at the right time, a fission explosion occurs. This method was used in the atomic bomb that destroyed Hiroshima at the close of World War II. |
|
Implosion |
For technical reasons, the gun-type assembly cannot be used in a plutonium bomb. Instead the method of "implosion" is used. A sphere of plutonium that is less than a critical mass can be made critical by compressing it to high density. The sphere is surrounding with high explosives in such a way that, when they are set off, the plutonium is compressed to a high density; it then becomes critical and explodes. (Implosion can be used with either plutonium or uranium.) The first atomic bomb ever exploded was a plutonium bomb tested in the New Mexico desert before the bombs were used in war. The bomb set off over Nagasaki, which ended the world war, was an implosion plutonium device. (Scientists were so sure of the gun-type device, that it was not tested before its use at Hiroshima.) |
|
Soviet espionage |
Implosion was a crucial and difficult engineering problem, overcome by the scientists at Los Alamos, where the bomb was built during the 1940s. It was also the secret obtained by the Soviet Union through espionage, which accelerated their development of the bomb. There is no doubt, however, that the Soviets would have obtained the bomb not too many years after the war in any case. |
|
The fusion bomb |
Before the fission bomb was even completed there was discussion at Los Alamos about the possibility of another kind of bomb based on fusion. This is a process, in a sense the opposite of fission, in which two light nuclei are brought together in a reaction which produces a heavier nucleus. The key reaction is the following: |
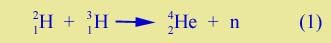 | |
| Here the two rare isotopes of hydrogen combine to form 42He, the common isotope of helium plus a free neutron. Because the helium nucleus is tightly bound (has a large binding energy), the reaction produces a great deal of energy. This is the energy of what used to be called the "H-bomb". | |
|
Ignition |
But remember that in order for a nuclear reaction, like (1) above, to occur, the nuclei must themselves be brought together at high energy. In the fusion bomb this high energy is produced by a fission bomb located at the core of the hydrogen fuel. The fission (plutonium) bomb is set off, and this provides the energy to "ignite" the hydrogen isotopes in the fusion bomb. |
|
Fission-fusion-fission |
A third step has been introduced in most of the nuclear weapons deployed today by the major powers: If the fusion bomb is surrounded by a coating of natural uranium (mostly 238U), the neutrons from the fusion reaction will set off fission in this uranium. It is not a chain reaction (in which the number of neutrons multiplies, starting with a small number), but simply a large flux of neutrons from the H-bomb causing fission of the natural uranium. This is a relatively cheap way of increasing the yield of the weapons. This system is sometimes called "fission-fusion-fission". |
|
Tritium |
A key ingredient in nuclear arsenals during the past 40 years, and today, is the isotope, 3H, also called "tritium". It is not found in the natural world. Tritium for nuclear weapons is produced in special military nuclear reactors, where it is formed by neutron absorption on naturally found hydrogen (in water). Tritium is radioactive, with a half life of 12 years, and so tritium must be continually produced and replenished in the weapon stockpile. |
|
Comparison to chemical explosives |
If we compare nuclear and chemical weapons in terms of the quantity of energy released per kilogram of material, the nuclear explosive is about 106 times as great. This factor of one million really reflects the fact that the nuclear force (the force that holds protons and neutrons together in nuclei) is about one million times stronger than the chemical force (the force that holds atoms together in molecules). |
|
Yield: Kilotons |
The energy release (called "yield") of a nuclear weapon is given in units called "kilotons" or "megatons". A kiloton is the energy released by 1000 tons of TNT. One ton of TNT made up the typical payload of the conventional (non-nuclear) bombs used during World War II. Atomic bombs, of the type used in the attacks on Japan, weighed in the range from 10 to 100 kilograms, and had yields in the region of 15 to 20 kilotons. |
|
Limit to the fission weapon |
Notice that it is not possible to make fission bombs significantly greater than this. If you think of the gun-type apparatus, you have to have two halves of the bomb, each of which is non-fissioning. If you wanted to make a fission bomb with a yield of 100 kilotons, the piece of uranium that made up half of it would be so large that it would constitute a critical mass itself, and it would spontaneously explode. On the other hand, there is no limit to how big you can make a fusion bomb. You just have to increase the amount of hydrogen fuel. |
|
Megatons |
Yields today are usually given in megatons, the equivalent of one million tons of TNT. The largest bomb ever exploded had a yield of 50 megatons; it was a test carried out by the Soviet Union. Today most nuclear weapons have yields of around 1 megaton. That is because there is relatively more destructive power in smaller bombs: e.g. more destruction caused by 10 bombs of one megaton than by one bomb of 10 megatons. |
KEY CONCEPTS
|