

|
| Some of the naturally found elements at the bottom of the nuclear table are unstable, and all of the artificially produced elements are unstable. What this means is that a given nucleus, after a certain period of time, spontaneously breaks into two parts. | |
Discovery |
Henri Becquerel discovered radioactivity in 1896, in experiments with compounds of uranium. He observed that photographic plates that were wrapped in black paper (keeping out the light) became exposed in the vicinity of uranium. Thus, some kind of "radiation" was capable of going through the paper, although it was stopped by a thick slab of metal. |
Types of Radioactivity |
Ernest Rutherford (in work done around the turn of the century) distinguished two components in this radioactivity, which he called alpha and beta. (Later the third type, gamma, was discovered.) He found that these radiations were bent in passing through magnetic fields, and he studied how effectively they penetrated matter. Alphas were less penetrating, and also bent less in magnetic fields. It was determined that alpha rays consist of a stream of charged particles which are the nuclei of helium, (even though the radioactive material contains no helium). Beta rays consist of a stream of electrons. In both cases the particles are fast-moving, meaning they move at some appreciable fraction of the speed of light (say, 1/30 of the speed of light or more). Nothing that fast (except light) had ever been observed before. Both alpha particles and beta particles are electrically charged. The third type of radioactivity, called gamma particles, can be thought of as similar to the particles that make up a beam of light (photons); but each gamma particle carries much more energy than a photon (although both travel at the speed of light). |
Alpha Decay |
As an example, 238U, the common isotope of uranium, decays slowly according to, |
 | |
| Element 90 is called thorium. Another example: | |
 | |
| Pu is plutonium. Why is a helium nucleus produced? The unstable nuclei, like uranium, are unstable because they have too many particles to hold together. You might think that they would decay by emitting protons or neutrons. That happens in some rare cases, but more commonly it is the alpha particle, largely because it is a grouping of protons and neutrons that is particularly stable. It is held together very tightly, more so than many of the other small nuclei like 32He, 31H, or 21H. | Neutron Generator |
Process (2) was used in the neutron generator that we used to have in the laboratory for our radioactivity experiment. Plutonium is strongly radioactive; a sample of plutonium is placed next to a piece of beryllium. Alpha particles continually stream from the plutonium, some of them hit beryllium nuclei, and according to a nuclear reaction discussed elsewhere, free neutrons are produced. Since there are risks in having plutonium in low security sites, it was replaced several years ago by americium, which is also an alpha particle source. |
Beta Decay | High energy electrons are found streaming from radioactive materials. These electrons do not come from the outer parts of the atom; they come from the nucleus. Two examples:
|
 | |
 | |
|
Pa is protactinium, Al is aluminum, and Si is silicon. Unlike the case of alpha decay, here the number of protons and neutrons is not the same before and after the process. In both (3) and (4) we have one more proton after than before. We also have one less neutron. The thorium nucleus has 90 protons and 144 neutrons; the protactinium nucleus has 91 protons and 143 neutrons. One of the neutrons in the thorium nucleus has disappeared, and in its place a proton has been created, and an electron has been created. The proton stays in the nucleus, making it a nucleus with different atomic number -- a different element. The electron doesn't stay in the nucleus. It flies out and is observed as beta radioactivity. In the laboratory you will study the beta decay of 28Al. |
|
Transmutation |
It was a basic dogma of nineteenth century science that atoms -- and hence the chemical elements -- could never be changed. The discovery of radioactivity in the closing years of the century, showed for the first time a process in which, according to our modern understanding of matter, the atomic number, and hence the chemical element, changes. This was called "transmutation of the elements" in medieval times and afterward. Alchemists were the precursors of chemists, who tried to convert lead into gold. |
Instability of the neutron | Note that in alpha decay we always have the same total numbers of protons and neutrons. Now we drop this restriction too. Protons and neutrons are not permanent; they can be created or destroyed. The basic process of beta decay can be written simply, |
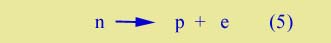
| |
Neutrinos | (Actually there is a third particle produced on the right side of (5) -- and in the beta decays discussed above. It is called a neutrino. It plays an important part in some of the theories of modern physics, but not in what we will be considering about beta decay, so we will omit it from further discussion. Its interaction with other particles is extremely weak, and as result it was not discovered until 1948.) |
Creation and destruction of photons |
The idea of creating matter out of nothing sounds very weird, but by the time beta decay began to be studied in detail, scientists had learned some other things that made process (5) not so unbelievable. For one thing, light can be thought of as a stream of particles - photons. These photons have energy and they have mass, and they are created out of nothing. Emerging from the filament of an electric light bulb are newly created photons. And when light is absorbed on a piece of black paper, the photons are destroyed. |
E = mc2 | Furthermore, it is not quite right to say particles are created out of nothing. In order for a particle to be created there must be available energy in some form, and this energy is converted into the mass of the particle. Einstein's theory of relativity (1905) had shown that such inter-convertibility between mass and energy was a fundamental property of the physical world. |
The stability of matter | You might wonder, given process (5), how there can be any neutrons around at all - for example, the neutron in heavy hydrogen nucleus, 21H. Wouldn't they all have turned into protons and electrons? The answer lies in the concept of binding energy. For a free neutron, the decay (5) can occur because the neutron has more mass than the total of the proton and the electron. But the neutron inside a nucleus, such as 21H, is in a different situation. The mass of 21H is less than that of its component parts, because the nucleus has binding energy. If the neutron decayed into a proton plus an electron, you would have |
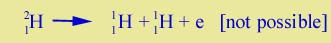
| |
|
But the binding energy of 21H is enough to make its mass less than the total of the three particles on the right. So the decay doesn't go. The same argument holds for most of the heavier nuclei that contain many neutrons: Nuclear binding energy prevents the neutron decay -- as in Equ. (5) -- from occurring. On the other hand, when a nucleus is beta radioactive, that means that its binding energy is not so great, its mass is not reduced very much, and thus one of its neutrons can decay, as in (3) and (4) above. | |
|
Radioactive Series |
Notice that decay (3) follows from decay (1). If we start with a uranium nucleus, it decays to thorium, and the thorium then decays to protactinium. Protactinium is also radioactive: it beta decays to become 234U. (Write this equation out to convince yourself that it makes sense.) This isotope of uranium then decays again. There is a series of decays, more than a dozen steps (some alpha, some beta, some gamma), which finally leads to one of the stable isotopes of lead. That is why when you pick up a sample of uranium you get a lot of different kinds of radioactivity -- because the sample really contains small amounts of all the different nuclei in the radioactive chain. |
| Marie Curie (in the early 1900s) analyzed uranium chemically, and discovered many new elements in this way. One was radium, one of the most intensely radioactive elements among the products of uranium. Another is radon, which is a gas, and so can accumulate in the air in houses, usually in basements. | |
| Units
| A number of different units are used to measure quantity of radiation. The curie is a measure of the amount of radioactivity in a given sample of material. It is approximately the radioactivity present in 1 gram of radium. The curie doesn't measure how much a radioactive dose a person will get, since that depends, among other things, on how close that person is to the source. One unit for the dose of radioactivity is the rad, which is defined in terms of a given amount of energy (of the decay particles) absorbed per kilogram of body weight. The rem is a unit similar to the rad, but defined so as to take into account the fact that the effect on the body depends not only on the energy of the radioactive particle, but on the type of particle (alphas, betas, neutrons, gammas). The lifetime dose an individual gets from the natural radioactivity in the environment (uranium and other minerals in the earth, cosmic rays from space) is about 7 rem. A chest x-ray gives about 0.03 rem. |
Health Effects |
Larger doses of radiation can have serious harmful effects on the human body, but there is still controversy about how serious the effect of a given dose will be. |
Half-life |
What is meant by the statement that a single nucleus, "after some time", will spontaneously decay? Nuclear decay occurs according to a concept called half-life. For any given nucleus there is a fixed time after which one-half of all the nuclei will have decayed. For example, the half-life of aluminum-28 is 134 seconds, meaning that if I start with a certain amount of this isotope, after 134 seconds half of it has decayed. After another 134 seconds (i.e. 268 seconds after the starting time) half of these remaining nuclei decay. Thus, after 268 seconds I am left with one-fourth of the original amount. After a total of 402 seconds I am left with one-eighth of the original amount, and so on. |
| At this point you should begin to work on Problem Set 4. | |
| |
|
Creation of the Elements |
Why are radioactive substances found naturally? It is believed that radioactive elements were created in stellar explosions before the solar system came into existence. The age of the solar system is about 4.6 billion years. Therefore, only nuclei with very large half-lives, approaching a billion or a few billion years, are still around now. Those with shorter half-lives all decayed away a long time ago. Artificially created elements are radioactive. Most have very short half-lives, a few days or a few minutes. But some last longer and these elements can be stored. Plutonium, with a half-life of 24,000 years, is an important example. |
KEY CONCEPTS
|