

|
|
Creation of neutrons |
The first question to ask about Equ. (1),
|
 | |
|
is, where do the neutrons (inside the 4He) come from? In fact, this question has a deeper significance than just the generation of solar energy. Since we believe the universe originated as a cloud of hydrogen gas, one has to ask where the neutrons came from which are part of the all the other elements that we find on earth, in the solar system, and beyond.
| |
|
Beta decay |
In all the nuclear processes that we've studied, the numbers of neutrons and of protons remain the same before and after, with one exception. That exception is beta decay , in which a neutron decays to a proton and and electron: |
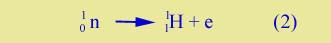 | |
|
Reverse beta decay |
But while beta decay was first discovered as a component in the radioactivity of uranium and other heavy elements, it has turned out that the process in Equ. (2) is just one of a set of related processes that have been predicted and discovered over the past 100 years. One of these converts a proton into a neutron: |
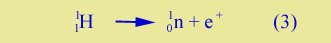 | |
|
Here e+ stands for a positron. It is a particle that has the same mass as the electron, but has a positive charge.
| |
|
Deuteron production |
Process (3) looks like a way in which a single proton can radioactively decay, but it cannot happen just that way, because the mass of the proton is less than the sum of the masses on the right side. Rather, it happens as a nuclear reaction in which a second proton participates:
|
 | |
|
Here 21H is the heavy isotope of hydrogen, also called the "deuteron". Be sure you see that process (4) is just the same as process (3), except that there is an extra proton on both sides. This proton and the neutron become bound together as an 21H.
The key is that the 21H has a binding energy; its mass is less than the mass of its component parts. And this difference is enough to make the total mass on the right less than that on the left. Hence, not only can the reaction go, but there is a net production of energy.
| |
|
Fusion |
Thus, not only is energy produced and a neutron created, but we achieve the first step in building up from a nucleus with one particle (11H) to a nucleus with two particles (21H). Two more steps build up to a nucleus with three particles, and then to a nucleus with four particles: |
 | |
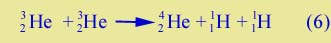 | |
|
Helium production |
Looking at Equs. (4), (5), and (6) together, you can see that we start with a hydrogen plasma, which is a gas of separated protons and electrons. Some protons interact and produce deuterons (21H's); these nuclei become part of the plasma and in time interact with protons (Equ. (5)) to produce 32He's; these also become part of the plasma, and in time interact with other 32He's to produce a 42He nucleus, with two protons left over.
|
|
The proton-proton cycle |
You should verify two things for yourself:
Because we start with six protons, but end up putting two back into the plasma, the net result is that four protons are taken out of the soup and produce one 42He. This sequence of three reactions is called the proton-proton cycle. |