Research Interests
We are a synthetic organic research group.
Catalysis: We develop new catalytic asymmetric reactions that we and others can use in the synthesis of natural products and drugs. Specifically, we have developed a number of cascade reactions, in which multiple synthetic steps are carried out in a single reaction flask, and which are catalyzed by organic molecules, known as organocatalysts. The methods we develop reduce the time, waste, and cost associated with the conventional organic synthesis of valuable synthons. Illustrated below are two examples of novel organocatalytic cascade reactions recently developed in the group.
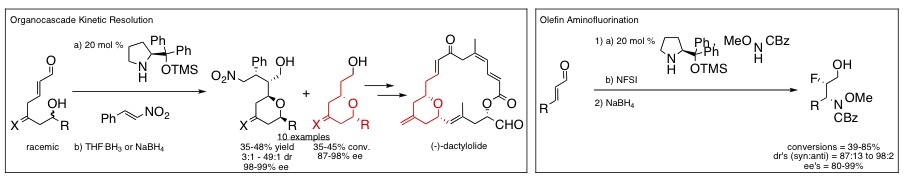
Medicinal chemistry: Our organocatalytic olefin aminofluorination reaction (illustrated above) generates chiral beta-fluoroamines, subunits increasingly incorporated in medicinal compounds. We will use this reaction to produce fluorinated analogues of small, bioactive amino compounds, with the goal of increasing the bioavailability and bioactivity of the amino compounds. Stay tuned for our results!
Facilities
We regularly use our polarimeter and GC, the departmental HPLC, FT-IR, and 200 and 400 MHz NMRs. In addition, we make use of CUNY’s mass spectrometry facility located on the Hunter College campus.
The chemistry department at Brooklyn College has other instrumentation, including LCMS, GCMS, and UV-VIS, that we use less regularly. In addition, CUNY has higher field NMR instruments as well as an X-Ray Crystallography facility that are available for our use.